ENRICHING LIVES
WITH SUPERIOR QUALITY
PHARMACEUTICAL PRODUCTS
OUR PRODUCTS
ADHERE TO HIGH INTERNATIONAL
ETHICAL STANDARDS AND GUIDELINES
CATERING CLIENTS
ACROSS THE GLOBE OPERATING
AS THEIR EXTENDED WING
ABOUT US
Abil Chempharma is a young company founded in the year 2000 and is based in Mumbai, India. We are in the business of manufacturing, marketing, distribution and exports of APIs, Intermediates & Finished Formulations.In a very short period Abil has developed huge acclamations for its work from its esteemed clients. Abil has successfully established a strong foothold in the Indian as well as overseas market.
Our wide range of activities can be summarized as :
Contract Manufacturing of a wide range of Finished Formulations
Exports of Finished Formulations to Semi-Regulated markets globally
New Product Development – driven by research, using advanced technology, regulatory approvals, quality led manufacturing
Official marketing partners and supplier of Active Pharma Ingredients for domestic and international market.
GET IN TOUCH
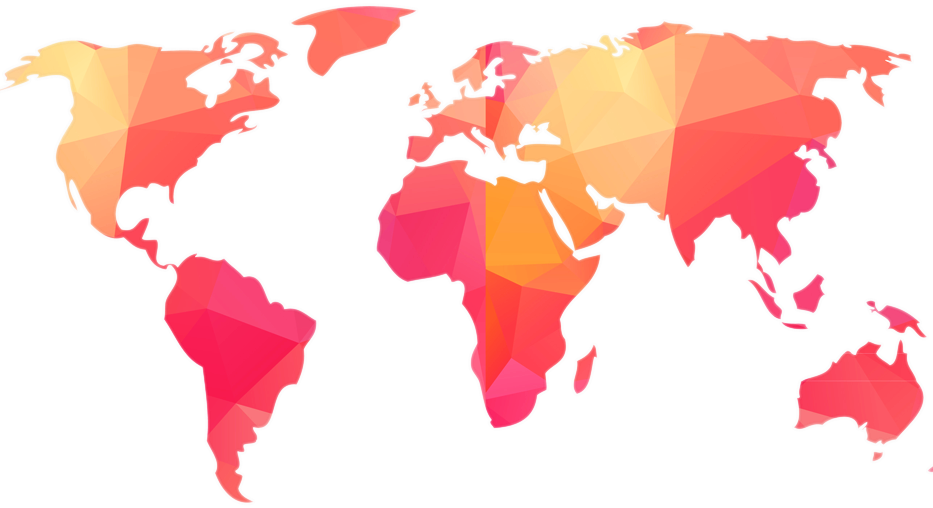
GLOBAL PRESENCE
ABIL has strong international presence with robust network of business partners & marketing offices strategically located across the world.
Over 100 products registered in various countries & 250 + products under registration in Africa, Central & Latin America, Middle East & SE Asia
We are searching for experienced business partners for distribution and marketing of our manufactured products.
For more information email on info@abilchempharma.in
Our Capabilities

Contract Manufacturing Services
The products we supply are manufactured in GMP certified sites and sites which have cleared multiple International audits.
New Product
We strive to constantly provide new products and innovative solutions that can have a positive impact on the lives of people

Regulatory Expertise
Our Regulatory team ensures that we deliver end-products which adhere to high quality international standards

Operational Efficiency
We deliver products or services to customers in the most cost-effective manner possible while still ensuring the high quality of our products, service and support.

Novel Actives
We are your ideal partner for sourcing high-quality and novel APIs.

Creating Customer Value
Creating and Delivering value is the Top Priority of our Business & Teams
Clients Testimonials
“ My association with ABIL goes a long way and it has become stronger with time due to their customer centric approach. When it comes to introducing new products and offering competitive prices ABILs team is the one to trust. ”

Finished Formulations
Domestic Customer
“ Happy with the International Business team of ABIL. Their understanding of the local market dynamics and support to grow our business and long term vision impressed me. I wish to add more products from their portfolio for my distribution business. ”

Distribution Business (Cambodia)
Owner
“ They have taken time to understand our requirements and then worked efficiently and timely to drive product registrations in our Country. They have a highly experienced BD and regulatory team and I would recommend potential business partners in Latin America to work with them. ”

Distribution Business (Nicaragua)
CEO
“ Excellent service. Operations team is very thorough in follow up and always delivering on timelines. They are our invaluable partners for formulations requirements. ”

VP Supply Chain
Domestic Customer of Finished Formulations
“ We work with ABIL for exploring & growing our Export business in African and Central American countries. They have been outstanding so far, consistently impressed with their marketing capabilities, network / connections and professionalism. I now feel they are my own team. ”

Finished Formulation Manufacturer
Managing Director
QUALITY APPROVED & CERTIFIED PRODUCTS
We only work with facilities that adhere to high international ethical standards and guidelines. All our marketed products are manufactured in GMP certified facilities and in compliance with international regulations and standards.
